Nvidia among investors in xAI’s $20 bln capital raise- Bloomberg
Introduction & Market Context
Lytix Biopharma AS (OB:LYTIX) presented its Q2 2025 results on August 28, 2025, highlighting significant progress in its innovative cancer treatment pipeline and improved financial performance. The company’s stock closed at NOK 9.04 ($0.86) on August 27, representing a 0.89% increase ahead of the earnings presentation. Lytix is developing novel immunotherapy treatments that address major shortcomings in current cancer therapies, which show good response in only 20-33% of patients.
As shown in the following slide, Lytix’s approach combines local killing of tumor cells with systemic immune activation, positioning the company to potentially transform cancer treatment outcomes:

Quarterly Performance Highlights
Lytix reported a substantially reduced net loss for Q2 2025 compared to the same period last year, primarily due to the reversal of prior accruals and lower R&D activity as certain clinical trials reach completion. The company highlighted several key developments during the quarter:
- Verrica Pharmaceuticals partnership is advancing LTX-315 toward Phase III in basal cell carcinoma (BCC) following a successful FDA End-of-Phase II meeting
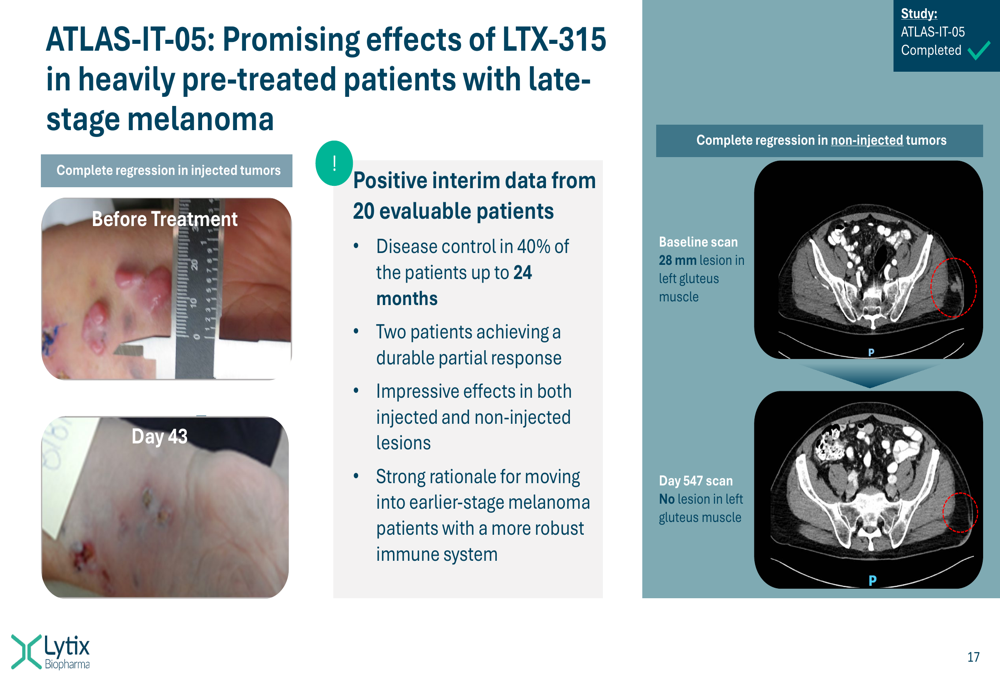
- ATLAS-IT-05 late-stage melanoma trial completed with disease control in approximately 40% of patients for up to 24 months
- NeoLIPA neoadjuvant melanoma study gaining momentum with one-third of patients enrolled and treated
- LTX-401 pipeline candidate showing strong preclinical results for deep-seated tumors
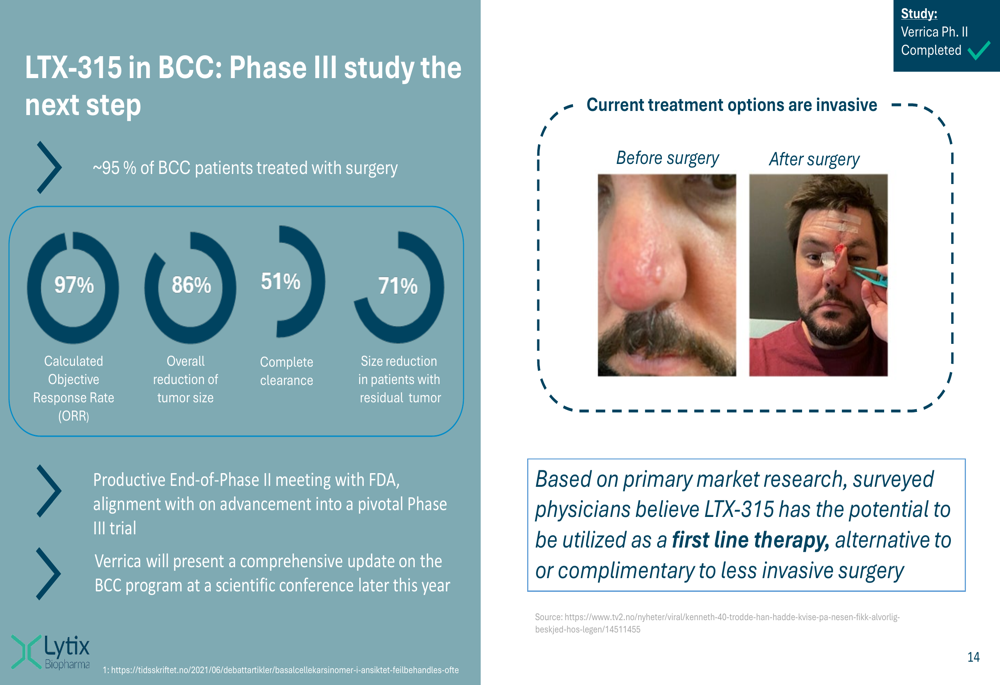
The following slide demonstrates the impressive clinical results for LTX-315 in basal cell carcinoma, which showed an 86% overall reduction in tumor size:
In the ATLAS-IT-05 melanoma study, Lytix reported promising effects in late-stage patients who had failed prior therapies, as illustrated in this slide:
Detailed Financial Analysis
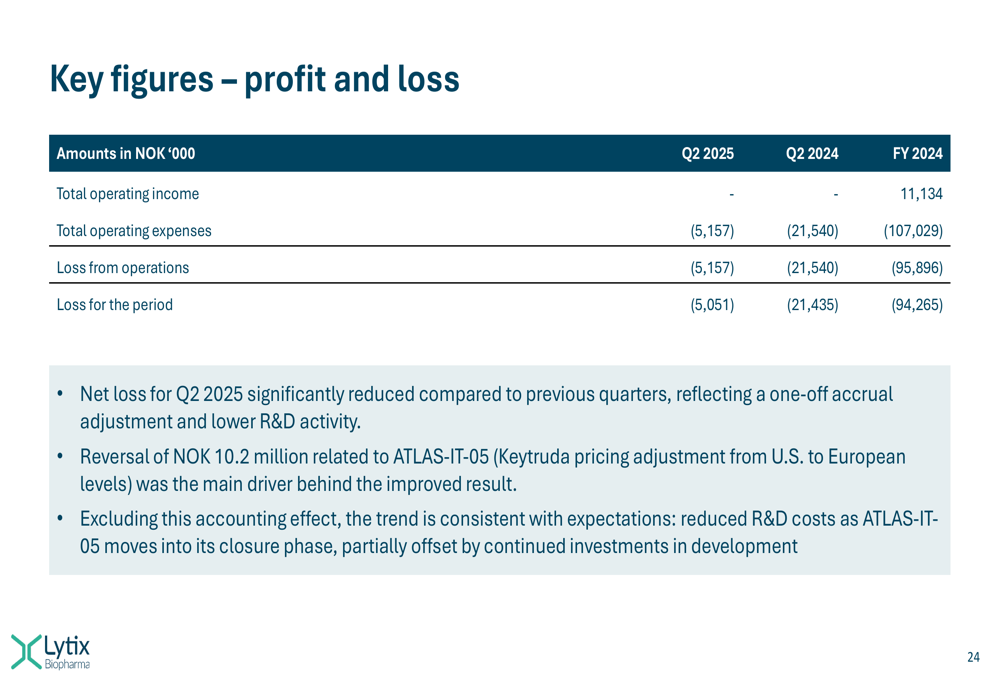
Lytix’s financial performance showed significant improvement in Q2 2025, with total operating expenses of NOK 5.16 million compared to NOK 21.54 million in Q2 2024. The company reported a net loss of NOK 5.05 million for the quarter, substantially reduced from NOK 21.44 million in the same period last year.
The following slide details the company’s profit and loss statement:
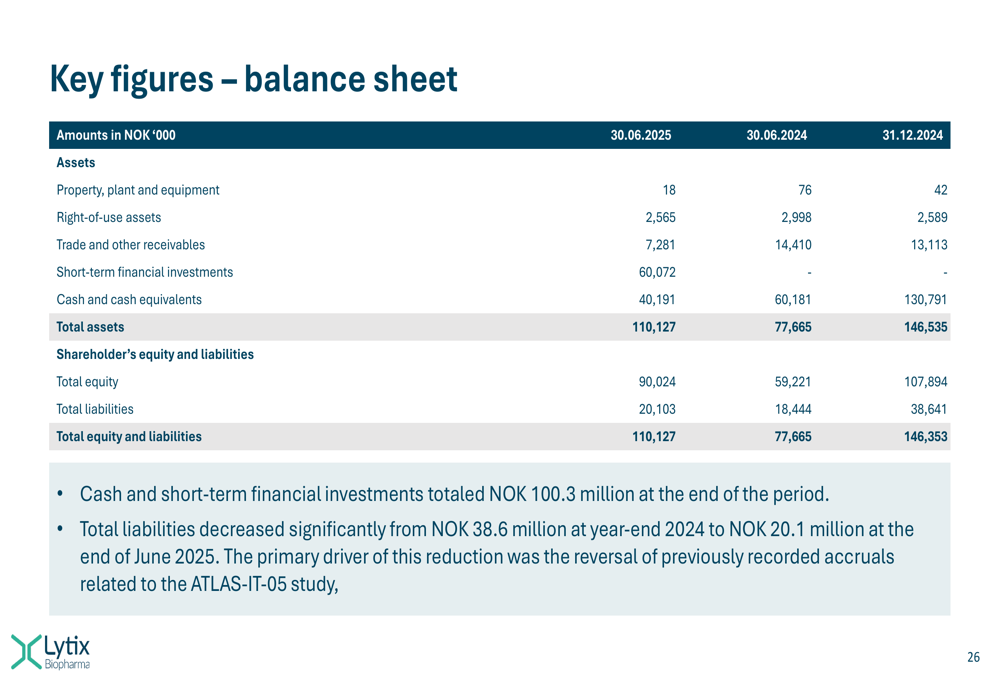
As of June 30, 2025, Lytix maintained a solid financial position with NOK 100.3 million in cash and short-term financial investments. Total liabilities decreased significantly from NOK 38.6 million at the end of 2024 to NOK 20.1 million, primarily due to the reversal of previously recorded accruals related to the ATLAS-IT-05 study.
The company’s balance sheet remains strong, as shown in this slide:
Management emphasized that the current cash position supports operations into 2026, providing flexibility ahead of key catalysts expected in the second half of 2025.
Strategic Initiatives & Pipeline Progress
Lytix is executing a multi-pronged strategy focused on three key areas: non-metastatic skin cancer, neoadjuvant melanoma and breast cancer, and deep-seated cancers. The company’s roadmap to create shareholder value is illustrated in the following slide:
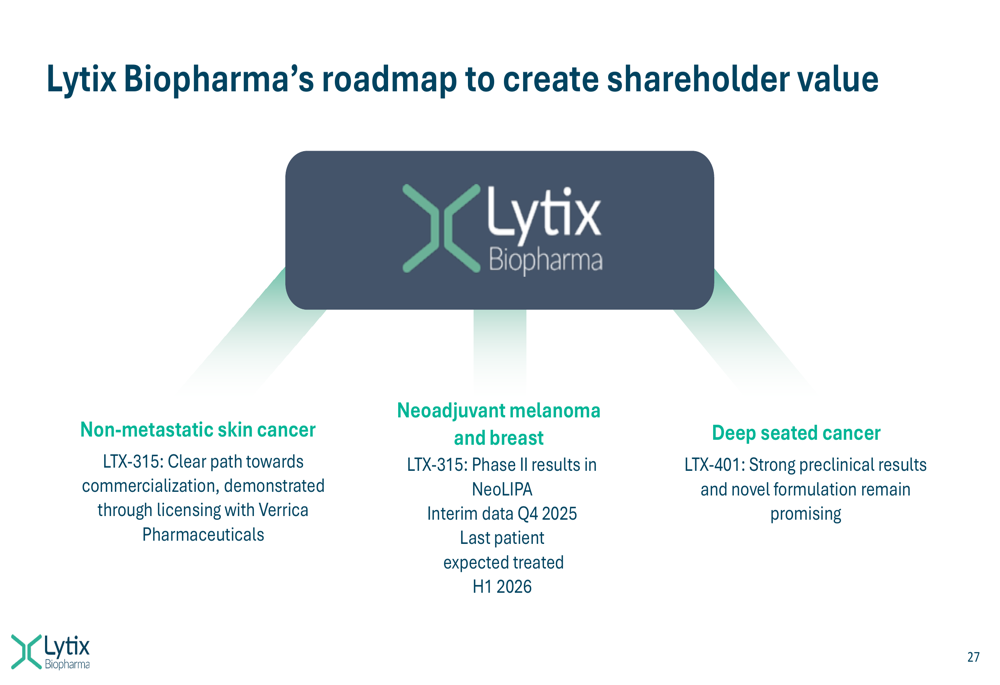
For non-metastatic skin cancer, Lytix is leveraging its partnership with Verrica Pharmaceuticals to advance LTX-315 toward commercialization. The Phase II results in basal cell carcinoma were particularly impressive, with a 97% calculated objective response rate and 86% overall reduction in tumor size.
In the neoadjuvant setting, the NeoLIPA study is evaluating LTX-315 in combination with pembrolizumab in early-stage melanoma patients. This represents a strategic shift toward treating patients with more robust immune systems, potentially increasing the likelihood of response to Lytix’s immunotherapy.
For deep-seated cancers, the company is advancing LTX-401, a small molecule oncolytic with similar mode-of-action to LTX-315 but with superior effects in liver cancer models. The company is reviewing the optimal timing and pathway for advancing LTX-401 to clinical development.
Forward-Looking Statements
Looking ahead, Lytix outlined several key milestones expected in the coming quarters:
- NeoLIPA interim results expected in November 2025
- Completion of ATLAS-IT-05 study in H2 2025
- Verrica preparing for pivotal Phase III trial in BCC, with additional immune/genomic data and program update expected in H2 2025
- Continued preparations for LTX-401 clinical development
CEO Øystein Rekdal emphasized the company’s transition toward commercialization, supported by promising clinical results and strategic partnerships. With a solid cash position extending into 2026, Lytix is well-positioned to execute its development strategy and potentially transform cancer treatment through its innovative immunotherapy approach.
The company’s cash flow statement provides additional insight into its financial trajectory:
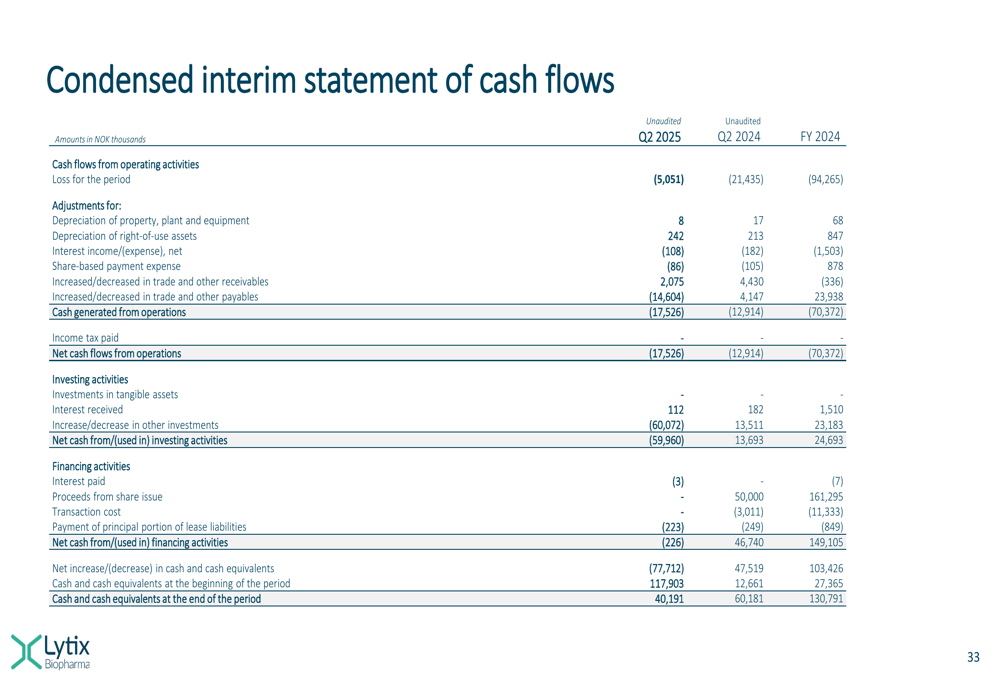
Investors will be closely watching the upcoming clinical milestones, particularly the advancement of LTX-315 into Phase III for basal cell carcinoma, which represents the nearest-term commercial opportunity for Lytix Biopharma.
Full presentation:
This article was generated with the support of AI and reviewed by an editor. For more information see our T&C.
